GxP in pharmaceuticals is a set of Good Practices; it ensures the quality and effectiveness of products for drugs, food, medical equipment, and other life-serving products as per regulatory standards.
Meaning of GxP in Pharmaceuticals:
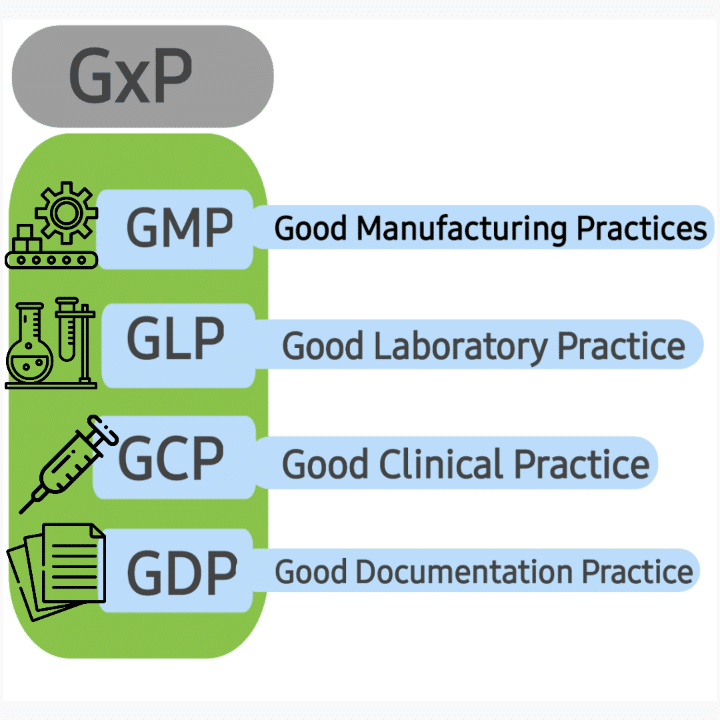
GxP is part of Good practice because it’s related to the manufacturing of life-saving products. GxP helped in controlling the product development process and ensuring the consistent production of quality products.
G= for Good, x= for a particular field, and P= for Practice
GMP- Good manufacturing practice
GLP- Good laboratory practice
GDP- Good distribution practice
GCP- Good clinical practice
GAMP- Good automated manufacturing practice
GACP- Good agricultural and collection practice.
Purpose of GxP
The purpose of GxP is to ensure that products are safe and effective for their intended use and that they are manufactured, quality tested, and distributed in a consistent and controlled manner. This is to help ensure the community’s safety and security of products. Some examples of specific GxP guidelines include Good Laboratory Practice (GLP), Good Clinical Practice (GCP), and Good Manufacturing Practice (GMP).
Why is GxP in industries important?
GxP in pharmaceuticals helps to control the potential risk to human health from the products they consume. GxP compliance is under the control of the regulator’s body and government agency. These different agencies monitored and controlled it by distributing certification and planning inspection and auditing. Other important areas include:
Data integrity:
Data integrity is a crucial element for GxP. Understanding the data is correct and recorded adequately with up-to-date and easily accessible and gives a high level of accuracy.
Documentation flow:
It is essential to do documentation, accountability, and traceability of a product throughout its life cycle. It demonstrates that the end product is manufactured and if any non-conformance is observed is identified and corrected simultaneously.
What is GMP in GxP:
GMP stands for Good documentation practice, also described as cGMP (current documentation practice) to ensure pharma products, medical equipment, and other regulated products are consistently produced in a controlled manner as per quality standards. It reduced the harm and potential risk to consumers.
The regulatory body like the US FDA ensures it by visiting different manufacturing sites and in vitro diagnostics to identify the follow-up of cGMP worldwide by the highly qualified FDA staff.
What is GLP in GxP:
GLP (Good laboratory practice) covers guidelines related to medicine, pesticides, cosmetics, veterinary drugs, and food additives.
The objective of GLP is procuring data integrity through GDP. GDP ensures documentation is safe and secure and readily available whenever required.GLP ensures that data obtained during different studies are accurate and reflect the exact result.
Good laboratory practice is reproduced by the QMS (Quality management system), and it ensures that every step taken consistently is appropriate.
What is GDP in GxP:
GDP (Good distribution practice) is significant for the sale, distribution, supply, and import of medicines. it states that nobody and company can directly do it without GDP practice. These guidelines ensure the supply chain of human medicine from manufacture to wholesale or pharmacy and then to the public.
The guidelines ensure the following functions:
- Storage of medicine and medical-related products in a specified condition.
- To prevent tampering and contamination.
- To manage complaints and recalls.
- Ensure authorized products enter the supply chain.
What is GCP in GxP:
GCP (Good clinical practice) is designed to regulate standards for conducting and reporting the different clinical trials on humans. The organization ensures accurate data and safety in clinical trials.
- Before conducting, a clinical trial, the possible risk must count against the expected gains. Risk factors must be less than expected benefits.
- Before starting, trials, procedures, and parameters must be authorized by the concerned review board and committee.
- The information related to trials must be shared with all personnel involved with proper training, education, and stage to be involved.
- All informational data must be recorded and stored at every stage.
- The records related to the subject must be confidential.
Frequently Asked Questions (FAQs)
A. Some examples of specific GxP guidelines include Good Laboratory Practice (GLP), Good Clinical Practice (GCP), and Good Manufacturing Practice (GMP).
A. GxP good x practice and GMP (Good Manufacturing Practice) are related, but they are not the same thing. GxP is a set of regulations and guidelines, in simple words, it provides direction to companies to produce a quality product that is effective to use, whereas GMP guidelines focus on maintaining clean and controlled manufacturing environments, ensuring proper equipment and facility maintenance, and implementing testing and procedure for quality control.
A. A GxP product refers to a product that is related to human health and is subject to good x practice guidelines (GxP) regulations. These products include pharmaceuticals, medical devices, diagnostic products, and others that are intended to be used for the community’s health.
A. Prescription drugs, over-the-counter medications, dietary supplements, Medical devices such as pacemakers, artificial joints, diagnostic imaging equipment, and many others Biologics such as vaccines and gene therapies, Cell and gene therapy products
Diagnostics and lab reagents.
A. Examples of GxP records include Records of manufacturing process documentation such as uses of types of RM, equipment, and results of testing materials. it also includes laboratory records, quality control records, Distribution records, training records, and complaint records.
A. In the pharmaceutical industry, non-GxP refers to products that are manufactured and are not subject to Good x Practice or any regulations. These products may not be related uses for human health, or they may not be regulated by agencies.
A. 1. Research and development activities that are not intended to directly support the development of new products or the improvement of existing products.
2. Manufacturing or production of non-pharmaceutical products such as chemical intermediates, laboratory reagents, and other Environmental monitoring activities that are not related to human health
3. Marketing, Sales, and distribution activities.
4. Administrative functions such as finance, human resources
A. 1. Documenting and following SOPs that outline processes for manufacturing, testing, and distributing products
2. Implementing employee training program and ensuring that personnel are qualified to perform their roles
3. Conduct regular internal audits to ensure compliance with regulations.
4. Establishing and maintaining records, such as production records, laboratory records, quality control records, and others.
5. Implementing a continuous improvement program.

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].
