SOP for Additional Packing Materials Request using format and ERP
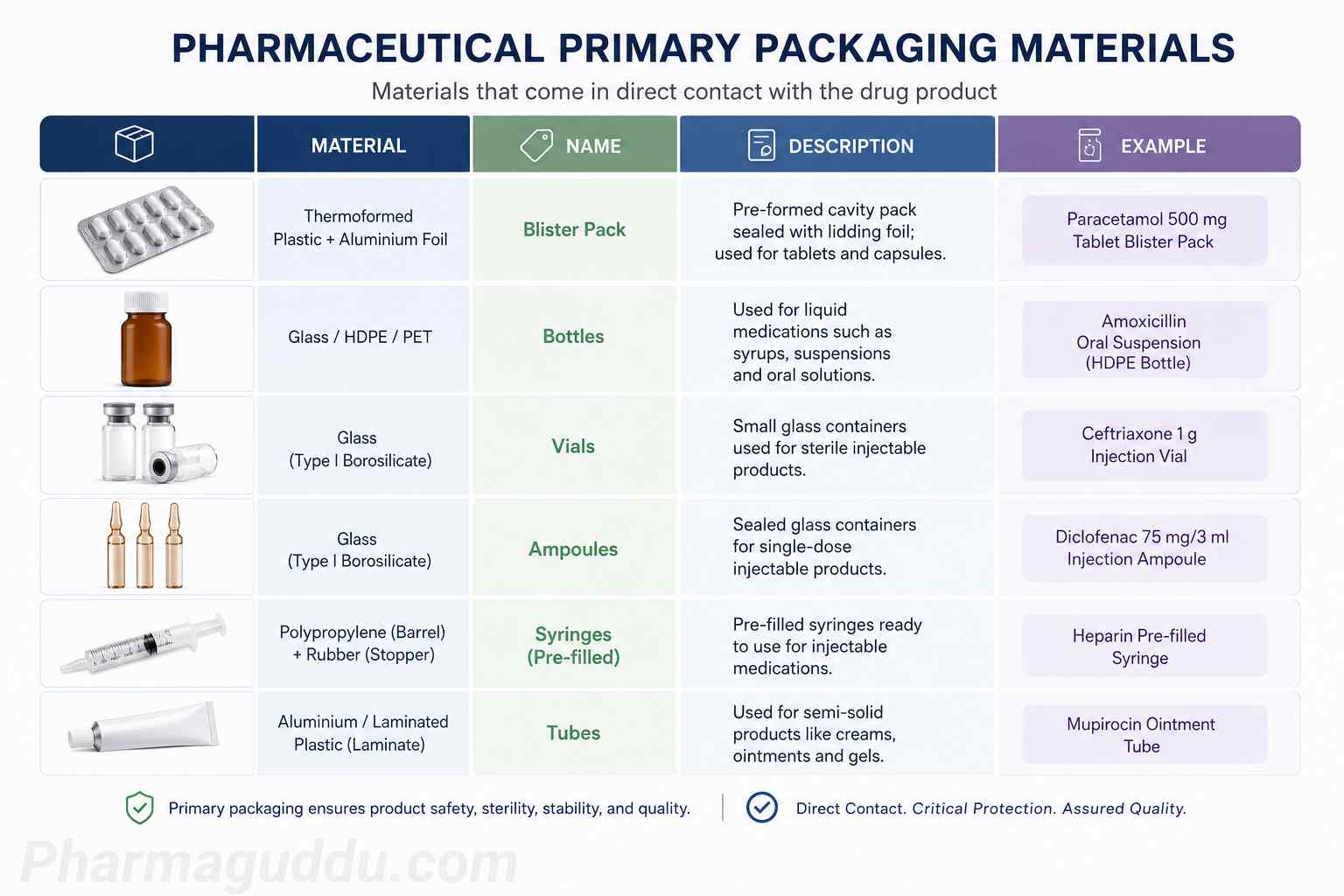
Download the SOP for Additional Packing Material in pharmaceutical manufacturing. Learn the GMP-compliant procedure for requesting, issuing, approving, and reconciling extra packaging materials