1.0 Objective: To lay down a Procedure for the Usage, Control, and Storage of Change Parts for liquid orals.
2.0 Scope: This procedure is applicable for the proper usage and control of change parts of the Bottle washing machine and Monoblock bottle filling & sealing machine in the liquid department.
3.0 Responsibility:
Operator, Officer, Executive – Production Department
Manager – Production Department
4.0 Procedure
4.1 Change Parts Usage.
4.2 In case of Product change over follow the below-mentioned pattern for using change parts which depend upon the bottle size.
4.2.1 Bottle Washing Machine Range:
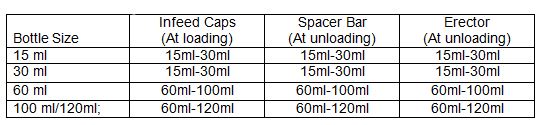
4.2.2 Carry out wet mopping of the machine followed by dry mopping with a lint-free cloth from outside.
4.2.3 Monoblock bottle filling and cap sealing machine Ranges:
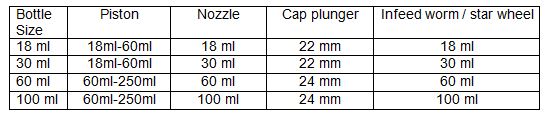
4.2.4 Ensure that all the used change parts are marked with their respective range as mentioned in steps No. 4.2.1 and 4.2.3
4.3 Control and Storage of Change Parts:
4.3.1 After completion of the batch dismantle the changed parts which are not required for the next product.
4.3.2 Check all the cleaned change parts for any damage. If any damage is found inform the concerned Executive/Assistant Manager.
4.3.3 Clean the changed parts and pack them.
4.3.4 Transfer all the dismantled, cleaned, and packed change parts to the accessory cabinet and put them in their designated place.
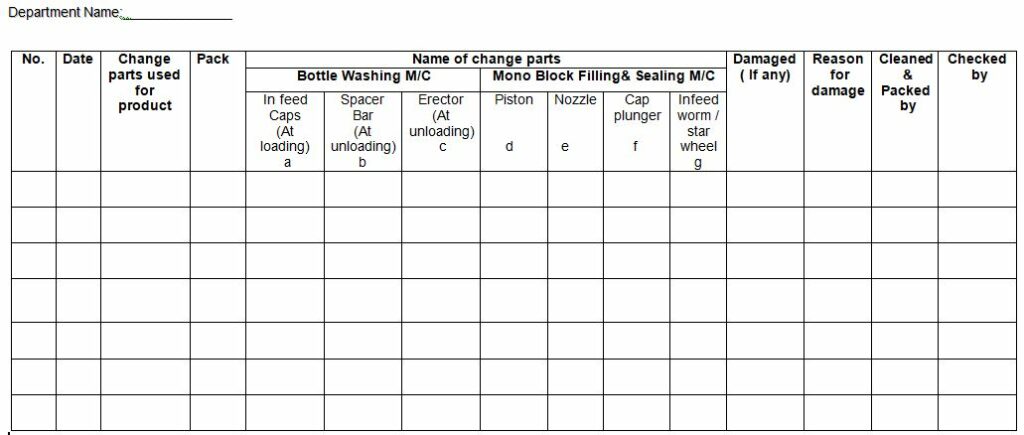
4.3.5 Record the usage and damage with the reason (if any) in the “Usage Record for Change Parts” as per Annexure – I.
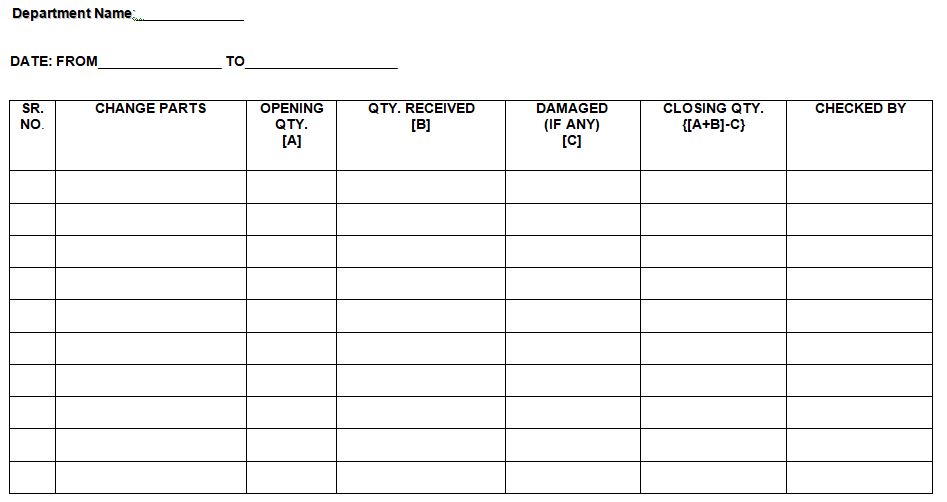
4.3.6 Physically check all the change parts at month end and Fill the “Change Parts Accountability Report” as per Annexure-II.
5.0 Abbreviations:
SOP: Standard Operating Procedure NO: Number
Annexure-1
Annexure- 2

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].
