Founder | Pharmaceutical Professional
✓ Reviewed by: Pankaj Sharma - Quality Control Specialist
Reviewed for Quality Control accuracy, laboratory practices, and technical relevance
📅 Last Updated: January 17, 2023
1.0 Objective: To lay down a procedure for the sanitization of drain points in pharmaceuticals.
2.0 Scope: The procedure is applicable to drain point sanitization in the production area at pharmaceutical companies.

3.0 Responsibility: Housekeeping personnel is to follow this procedure.
officers, Executives are responsible to implement this SOP.
Manager – Production Department responsible for Overall compliance with this SOP
4.0 Procedure:
4.1 Sanitization Of Drain Points in general areas
4.1.1 Clean the drain with a nylon scrubber followed by purified water.
4.1.2 Prepare the disinfectant/ sanitizing solution as per SOP ‘Preparation of disinfectant/ sanitizing solution‘.
4.1.2 Pour 500 ml of 2.5% v/v Dettol/ savlon solution into the drain and allow the solution to remain for 15 minutes in the drain points.
4.1.3 Change the disinfectant solution daily.
4.1.4 Sanitize the drain point at the end of the day.
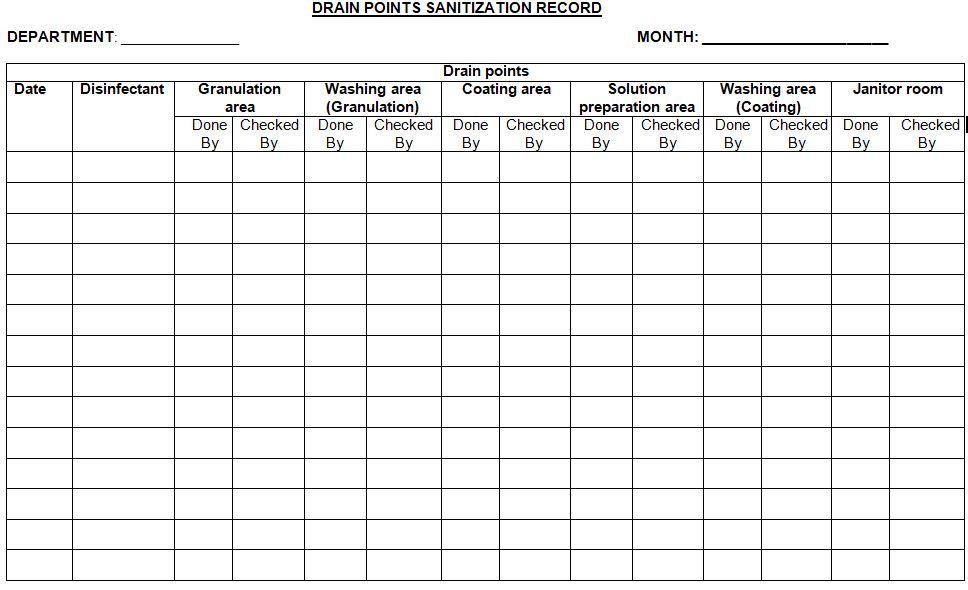
4.1.5 Record the sanitization activity in the given in Annexure –I.
4.2 Disinfection In the Sterile Section
4.2.1 The drain points covering the mesh shall be cleaned with a nylon scrubber.
4.2.2 Cleaned the drain points with a nylon scrubber by opening the mess to avoid water clogging.
4.2.3 Pour 500 ml of 2.5% v/v Dettol/ savlon solution into the drain and allow the solution to remain for 15 minutes in the drain points
4.2.4 Close the drain by the mesh & cover it.
4.2.5 Record the sanitization activity in the given in Annexure –I.
4.3 Precautions
4.3.1 Use Disinfectant Savlon/Dettol every alternate week.
Related: Cleaning, sanitizing in Pharmaceuticals.
5.0 Abbreviations:
QA: Quality Assurance
SOP: Standard Operating Procedure
v/v: Volume by Volume
ml: Milliliter
Annexure-1

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].
