Founder | Pharmaceutical Professional
✓ Reviewed by: Pankaj Sharma - Quality Control Specialist
Reviewed for Quality Control accuracy, laboratory practices, and technical relevance
📅 Last Updated: April 25, 2023
High-Performance Liquid Chromatography (HPLC) is used in pharmaceuticals for the analysis of drug mixture and their components. HPLC is reverse-phase Liquid Chromatography. This technique is used for thermal labile, high molecular weight peptides, and non-volatile compounds.
The main purpose of HPLC is used to examine the mixture for its identification, impurities, and purification.
The mixture of compounds that can be retained by a column can be separated by a column. HPLC separations have been achieved based on the difference in polarity, size, shape, charge, a specific affinity for a site, stereo, and optical isomerism.
For HPLC, The pressure of 1000-6000 psi for these double reciprocating pumps is used, which produced a pulse-free stream of mobile phase.
HPLC can be operated by both isocratic and gradient elution. Particle is generally 5um for analytical columns. Generally, a guard column is there before the main column, which has a particle size of 30um, to filter the impurities.
The sample is introduced to a column by either autosampler, which uses an injection needle. Take a sample from the sample vial placed in the sample rack, which is a part of the High-Performance Liquid Chromatography unit, or the sample can be introduced manually via injection in the column.
HPLC system components
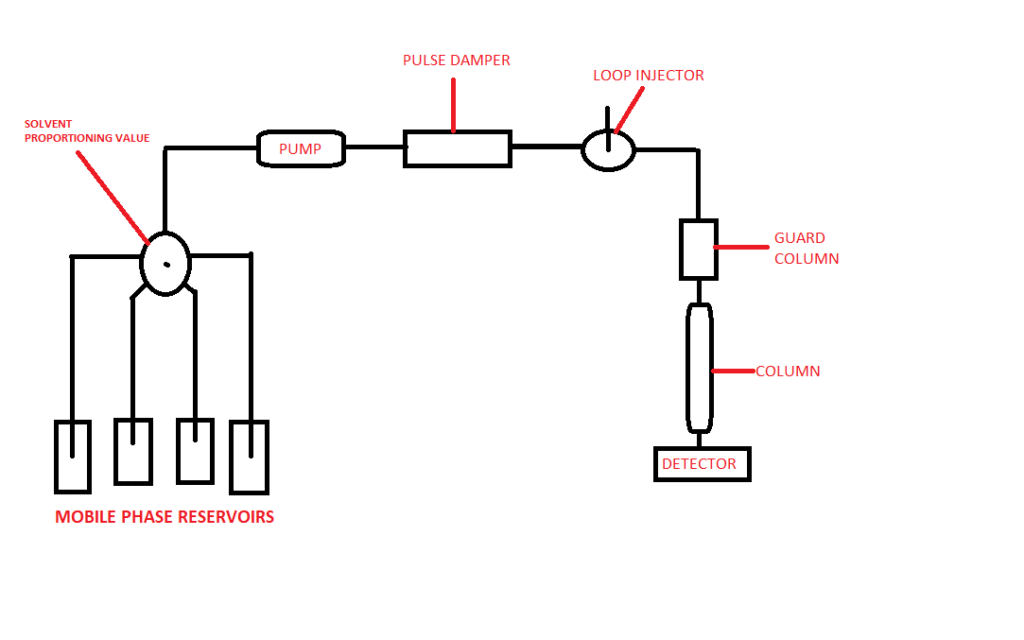
- Mobile Reservoir
- Reciprocating pump to build the pressure (1000 psi-6000 psi)
- Autosampler and injection system
- Column placed in the oven
- Detector
- Sample collector
Types of HPLC based on the component and their separation:
- Normal Phase columns
- Reverse phase columns
- Ion exchange columns
- Size exclusion columns
1. Normal Phase columns: It has a more polar stationary phase than the mobile phase due to packing materials being silica, which has more polar in nature. Water is more polar than silica, e.g., for normal phase columns. Water should not be used. Methylene chloride, hexane, and chloroform, or this combination with diethyl, is either used as a mobile phase. If sample components have more polarity, then these polar components strongly interact with the stationary phase, which is polar. So, elutes slower. If sample components have less polarity, then these less polar components weekly interact with the stationary phase, which is polar.so, elutes early.
2. Reverse Phase columns: it has a non-polar or less polar stationary phase than the more polar mobile phase. Bonded hydrocarbons like C 8 & C 18 and other non-polar hydrocarbons are used as stationary phases in reversed-phase columns. An aqueous organic solution like water-methanol or water acetonitrile mixture is used as a mobile phase. The separation of components occurs based on the polarity nature. It will be exactly opposite to the normal phase High-Performance Liquid Chromatography (HPLC) column, e.g., it is called reverse phase chromatography.
3. Ion exchange column: easily ionizable components can be analyzed by using ion-exchange columns. The stationary phase in these columns remains acidic, or the basis has a negative or positive charge, while the mobile phase is polar. Separation is based on the attractive ionic force between molecules and charged stationary phase due to the exchange of ions during the separation of components, known as ion-exchange chromatography.
4. Size exclusion columns: Porous stationary phase is used in size exclusion columns, separation of the components according to their size. Polysaccharides and silica is used as a stationary phase in these columns. Small components or molecules penetrate the porous of the stationary phase. Then large sample molecules penetrate partially into porous, e.g., large sample molecules will elute fastly, and small molecules will eluate later.

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].
