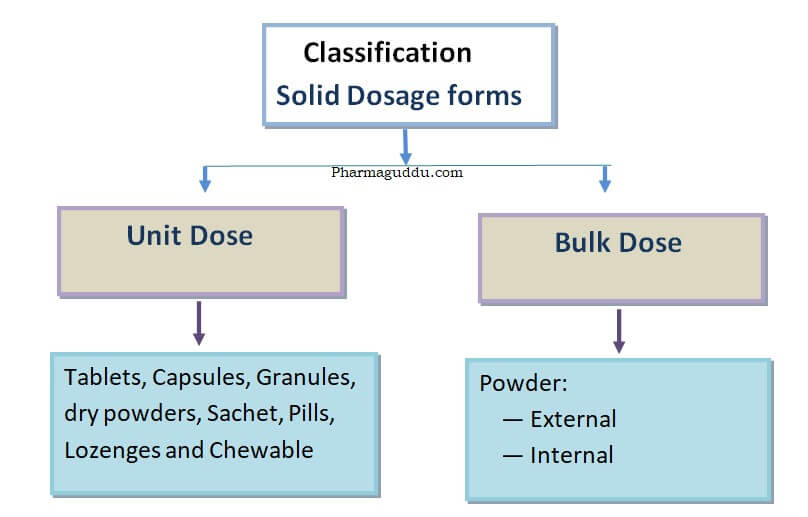
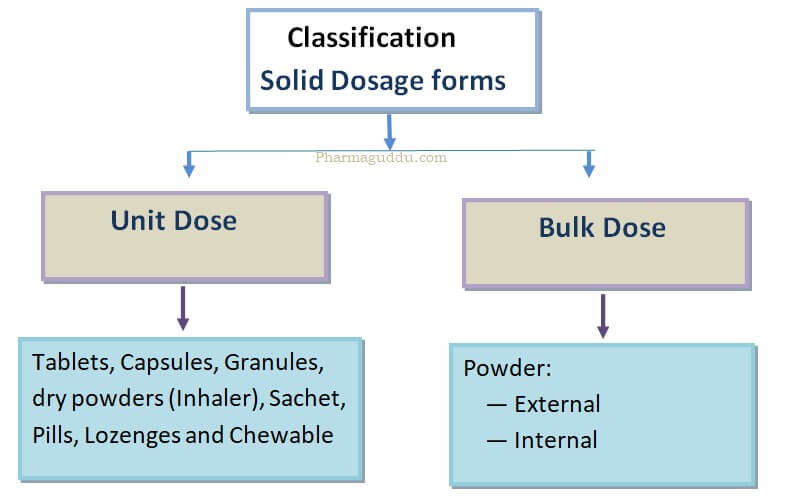
Tablets are solid dosage forms that are manufactured worldwide in the pharmaceutical industry. These are manufactured by molding or by compression method. Tablets or capsules come in the solid dosage form categories. Examples of solid dosage forms include tablets, capsules, powders, dry powders, and sachet.
What are Tablets in Pharmaceuticals?
Tablets are unit, solid dosage forms that contain one or more Active pharmaceutical ingredients. Tablets are administered orally. So it is also called solid oral Dosage form.
Ingredients or excipients used in Tablets:
Tablets contain additives (binders, diluents, disintegrating agents, lubricants, glidants, and other coating materials for the coating of tablets). Other materials are also added to form a complex with active ingredients after administration. Organoleptic agents (permitted coloring, flavoring, and sweetening agents) are also added whenever required.
Tablets or solid dosage forms are swallowed with water milk, and other suitable liquids. Other forms are formulated for chewed, swallowing, dissolved, and dispersed in water before administration. Some are retained in the mouth for the release of drugs.
Dosage forms like Pessaries and implants administered other than the oral route are also presented in tablet form or solid dosage forms. But these require special requirements for the formulation.
A General Property Tablets:
- The tablets must withstand physical and mechanical manufacturing, packing, shipping, and dispensing.
- The drug content of tablets must be bioavailable, which means they must be able to release their contents and active materials in a predictable manner.
- Tablets must be uniform in weight and drug content.
Types of Tablets in Pharmaceuticals:
- Core tablets or uncoated tablets
- Coated tablets: a. Film-coated tablets b.Sugar-coated tablets
- Enteric-coated tablets
- Dispersible tablets
- Modified release tablet
- Prolonged released tablets
- Effervescent tablets
- Lozenges and sublingual tablet
1. Core Tablets or Uncoated Tablets:
These are compressed tablets, maybe a single or double layer. These tablets are designed to provide rapid disintegration in the gastric fluid of the stomach.
Uncoated tablets are formed by the compression of active pharmaceuticals with excipients like (bulking agents, disintegrates, binders, lubricants, and some coloring and sweetening agents). These help tablets withstand during manufacturing, storage, and use.
The addition of coloring and flavoring agents to uncoated tablets (other than multilayer tablets) is not official unless permitted in the individual monogram of official books and pharmacopeias.
Examples of compressed tablets for oral, buccal, sublingual, or vaginal administration. paracetamol 500 mg and clotrimazole tablets (for vaginal insertion)
2. Coated Tablets:
Coated tablets are categorized into two types; a. Film-Coated Tablets and b. Sugar-Coated Tablets
A. Film-Coated Tablets:
These are tablets coated or sprayed with a thin layer of polymers(hydroxypropyl methylcellulose and hydroxypropyl cellulose) or a mixture of polymers, e.g., Eudragit E100, which are responsible for forming a thin layer of film on compressed tablets.
The thickness of such coating is usually between 20-100 um. The coated tablet is designed to break to expose the core tablets at a desirable location in the gastrointestinal tract.
Examples of film-coated tablets are Erythromycin, Amitriptyline, Diclofenac potassium USP 100 mg, and valsartan 320 mg film-coated tablets.
B. Sugar-Coated Tablets:
The sugar solution used with other additives for coating is called sugar coating. It has the following steps:

- Sealing of tablet core: Sealing of tablets is done by using polymers such as (shellac, cellulose acetate phthalate, and polyvinyl acetate phthalate). They protect the core of tablets from moisture.
- Subcoating: Subcoating of tablets is done by adding a bulking agent, e.g., calcium carbonate or talc, in combination with sucrose solution.
- Smoothing: Use sucrose syrup to remove the dull surface formed in step 2 stages.
- Coloring: For coloring tablets, Use Titanium-based pigments.
- Polishing: For the polishing of tablets, commonly, beeswax or carnauba wax is used.
- Printing: Indelible ink is used for printing on sugar-coated tablets.
Importance of Sugar-Coated Tablets:
Sugarcoating is concentrated sugar that helps mask objectionable masks and odors, increases stability, and improves patient compliance to unit solid dosage forms. It required more time and expertise in the coating process.
Examples of sugar-coated tablets include ferrous sulfate 200mg, ibuprofen 200 mg, conjugated estrogen 625mcg, and mebeverine hydrochloride 100mg.
3. Enteric-Coated Tablets:
Enteric-coated tablets are covered with one or more layers of the coating solution. The purpose of an Enteric coating is to provide resistance to the gastric fluid in the stomach.
Finally, drugs release their active ingredients in the intestinal fluid. These types of coating are aimed to prevent the formulation from gastric fluid OR gastric acid in the stomach. so enteric coating helps Drugs release the component directly inside the intestinal region.
Polymers like CAP( cellulose acetate phthalate), and hydroxypropyl methylcellulose succinate which is insoluble at low PH, but soluble at high PH. An example of enteric-coated tablets is diclofenac sodium delay release 100mg tablets.
4. Dispersible Tablets:
Dispersible tablets are uncoated tablets that produce a uniform dispersion in water and may contain permitted coloring and flavoring agents.
5. Modified Release Tablets:
These kinds of medications give immediate dose necessities for traditional therapeutic responses, followed by a gradual release of drugs in an amount sufficient to maintain the therapeutic response for specific extended periods of time, usually 8-12 hours.
6. Prolonged Release Tablets:
Prolonged-released tablets are also called sustained-release tablets and extended-release tablets. These are designed to release their medication in a predetermining manner over a prolonged period. Examples of Prolonged released tablets are Divalproex sodium extended-release tablets.
7. Effervescent Tablets:
Effervescent tablets release carbon dioxide after mixing with water because they contain acidic substances like, carbonate or bicarbonates, and act as a disintegrator which either produces a drug suspension or an aqueous solution.

These tablets are prepared by compressing active ingredients with a mixture of sodium bicarbonate and an organic acid such as citric and tartaric acid. An example of Effervescent tablets is vitamin-C and calcium tablets.
8. Lozenges and Sublingual Tablets:

Lozenges and sublingual tablets are used in the mouth, and Lozenges and sublingual tablets are usually uncoated and formulated to chewed or to effect a slow-release and local action of active ingredients(lozenges) or the release and absorption of active ingredients under the tongue (sublingual tablets). An Example of Lozenges and sublingual tablets is Nitroglycerin tablets for Angina pectoris.
Advantages and Disadvantages of Tablets:
Advantages of Tablets:
- Most stable with respect to physical, chemical, and microbiological attributes.
- Easy to handle, use, and carry out with an attractive and elegant appearance
- Easy to swallow, and production does not require any additional processing steps.
- Unit dosage form with accurate, stable dose.
- Protect medicaments from atmospheric conditions like air, moisture, and light.
- Large production is easy with low manufacturing costs as compared to other solid dosage forms. The unpleasant tests may mask by sugar coating.
- It can be formulated with two or more drug substances, even physically and chemically incompatible with avoiding taking multiple tablets at one time.
Disadvantage of Tablets:
- Increase product loss due to multilevel processes during manufacturing.
- A drug that is amorphous and low density or floppy powder ( cefpodoxime Proxetil), the character is difficult to compress during the compression stage.
- The Hygroscopic drugs are not suitable for compression. Because it takes moisture and causes a problem during the compression, sensitivity to oxygen drugs may require a special coating.
- The Absorption of the drug depends on physiological factors, such as gastric empty time, and it varies from one patient to another.
- To remove the bitter and unpleasant test coating is required, which causes more production costs.
- Some tablets may cause a problem in availability.
Solid Dosage Forms Examples: Solid dosage forms examples include Tablets, Lozenges, suppositories, and capsules.
Standard and Quality Control of Solid Dosage Forms: Tablets

This test is not applicable to coated tablets other than film-coated tablets. Tablets are required to comply with the test for uniformity of content for all active ingredients.
Uniformity of weight:
This test is not applicable to coated tablets other than film-coated tablets. tablets are required to comply with the test for uniformity of content for all active ingredients.
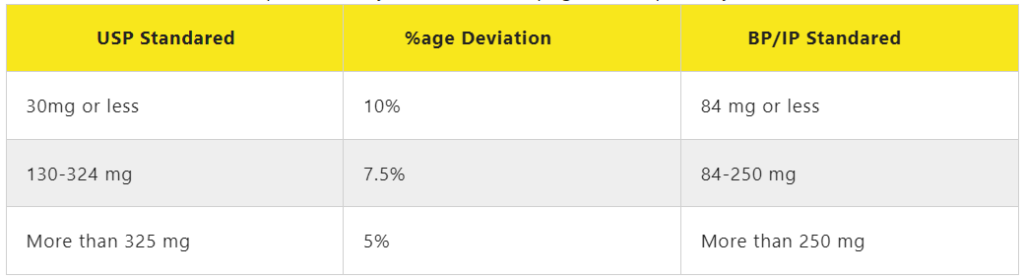
Weigh 20 tablets selected randomly and calculated the average Weight. not more than two of individual weights deviate from average weight by more than the percentage shown in a table and non-deviate by more than twice that percentage.
Uniformity of content:
This test is applicable to a tablet that contains less than 10 mg or less than 10% w/w of active ingredients. Tablets containing more than one active ingredient perform the test for every active ingredient that corresponds to the aforementioned conditions. The test for uniformity of contents isn’t applicable to tablets containing multivitamins and trace elements.
To determine the content of active ingredients in each of the 10 tablets taken at random. It complies with the test if, Not more than one (9 tablets out of 10) of the individual value obtained is outside the limits (85% to 110%) of the average value. None is outside the limits (75 to 125%) of the average value.
If two or three of the individual values are outside the limits (85 to 115%) of the average value and none is outside the limits (75 to 125%). Again repeat the test using another 20 tablets.
It complies with the test if the total sample of 30 tablets (10+20) is not more than three of the individual values outside the limits (85 to 115%). And none is outside the limits (75 to 125%) of the average value.
Disintegration test of Tablets:
This test is not applicable to modified-release tablets and tablets for use within the mouth. The Disintegration test apparatus must have a water medium at 37 ± 2℃. The basket moves up and down through a distance of 5-6 cm at a frequency of 28-32 cycles/ minutes.

A. Uncoated Tablets:
Disintegration within 15 minutes.
B. Coated Tablets:
Operate the disintegration apparatus for 30 minutes for film-coated tablets and 60 minutes for other coated tablets unless otherwise directed in the individual monogram.
For coated tablets other than film-coated tablets, if any of the tablets have not disintegrated. Repeat the test on a further six tablets, replacing the water in the vessel with 0.5M hydrochloric acid. The tablets comply with the test if all six tablets have disintegrated in the acid medium.
C. Enteric-Coated Tablets:
If the tablet has a soluble external coating, submerge the basket in the water at room temperature for 5 minutes. Suspend the assembly in the beaker containing 0.1M hydrochloric acid and operate with the disc for 120 minutes; remove the assembly from the liquid. No tablet shows signs of cracks that would allow the escape of the contents of this integration, apart from fragments of the coating.
Replace the liquid in the beaker with mixed phosphate buffer pH 6.8 at a disc to each tube and operate the apparatus for a further 60 minutes. Remove the assembly from the liquid. The tablets pass the test if all six have disintegrated.
D. Dispersible and Soluble Tablets:
Disintegrate within 3 minutes when examined by the disintegration test for tablets and capsules, using water at 24 to 26℃.
E. Effervescent Tablets:
Place one tablet in a 250-ml beaker containing water at 20-30℃. Numerous gas bubbles evolved when the evolution of gas around the tablet or its fragments ceased; the tablet shall have to integrate, being either dissolved or dispersed in the water so that no agglomerates of particles remain.
Repeat the operation on five further tablets. The tablets comply with the test if each of has six tablets disintegrates in the manner prescribed within 5 minutes unless otherwise stated in the individual monograph.
Dissolution Test for Tablets:
DT is used in all stages of the development of drug release and stability testing. It is an analytical method used for testing the amount of time required for drug substances in tablets to go into solution in a specific set of conditions. It provides the physiological availability of substances.
The dissolution medium for each drug is available in individual monograms, for basic drugs acidic medium is used, e.g., 0.1 M Hydrochloric acid while the alkaline medium is used for acidic drugs, e.g., alkaline buffers, water is used for non-ionizing molecules.
The dissolution test is performed at 37± 2° C. The following is the assembly part used within the dissolution test apparatus with their rotation speed and Solid dosage form to be tested.
Basket– for capsules and operated for 100 RPM
Paddle– for tablets and operated at 50 RPM
Reciprocating cylinder – At 635 DPM (dips per minute) for bead-type modified release dosage forms
Flow cell– for modified release dosage form with limited solubility.
Paddle over disc and cylinder– for transdermal dosage forms
Reciprocating disc– for non-disintegrating type oral modified release dosage form.
The Uniformity of Color:
The uniformity of color and gloss on a tablet surface is measured by a micro reflectance photometer.
Thickness:
Thickness is an important Quality test for tablet packaging. A very thick tablet affects packaging either in blister or plastic container tablets. Thickness is determined by a micrometer or sliding caliper. Tablet thickness should be controlled within a ±5% variation of the standard value.
Tablet Hardness:
The test measured the crushing strength property of tablets, defined as the compression force applied diametrically to a tablet that just fractures it.
Among a large number of measuring devices, the most favored once are used for solid dosage forms are the Monsanto hardness tester, Pfizer tester, and strong comb hardness tester; all are manually used. Other measuring devices, the Erweka hardness tester, and the Schleuniger hardness tester are motor-driven.
Friability Test of Tablets:
The Friability test for tablets is important because, During coating, packaging, and transport, the tablets may be subjected to a tumbling motion. That does not serve enough to break the tablet surface. For examination, tablets are subjected to a uniform tumbling motion for a specified time to calculate weight loss.
The plastic chamber revolves at 25 rpm. Normally preweighed tablet sample is placed in a friability which is then operated for 100 revolutions. The tablets that are less than 0.5-1% of their weight are generally acceptable.

Summary:
Tablets are solid forms widely produced in the pharmaceutical industry through molding or compression methods. Some examples of Oral solid dosage are tablets, capsules, powders, dry powders, and sachets. Tablets are unit, solid dosage forms containing active pharmaceutical ingredients, administered orally. They may include various excipients like binders, diluents, disintegrating agents, and coatings.
Tablets can be swallowed, chewed, dissolved, or dispersed in water. Tablets Types include core tablets, coated tablets (film-coated and sugar-coated), enteric-coated tablets, dispersible tablets, and more. Each type serves specific release and administration purposes. Tablets must meet criteria for weight uniformity, drug content, and disintegration time. Various advantages, like stability and ease of handling, coexist with disadvantages such as increased product loss. Quality control involves tests for weight uniformity, content, disintegration, dissolution, color, thickness, hardness, and friability.

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].