Preparation and Standardization of 0.01 M Sodium Tetraphenylborate (C6H5)4BNa) solution and calculating formula in Pharmaceutical labs.
Name: 0.01 M Sodium Tetraphenylborate (C6H5)4BNa)
Reagents: Sodium Tetraphenylborate
Sodium Hydroxide
Aluminium Hydroxide
Sodium Chloride
Cetylpyridinium Chloride
Potassium Chloride

Method of Preparation:
- First, mix 3.5 grams of sodium tetraphenylborate in 50 ml of water.
- Shake this with 0.5 grams of Aluminium hydroxide gel for about 20 minutes.
- Then, add 250 ml more water and 16.6 grams of sodium chloride.
- Let it sit for 30 minutes. After that, filter it. Add 600 ml of water and make sure the pH is between 8.0 to 9.0 using 0.1 M sodium hydroxide.
- Finally, dilute it to 1000 ml with water.
Method of Standardization:
- Before using, check its exact concentration in this way:
- Dissolve 7 mg of potassium chloride (previously dried at 150°C for 1 hour) in 5 ml of acetate buffer pH 3.7 and 5 ml of water.
- Add 15 ml of the sodium tetraphenylborate solution and let it stand for 5 minutes.
- Then filter it through a dry, sintered glass filter.
- To 20 ml of the filtrate, add 0.5 ml of bromophenol blue solution.
- Titrate the excess sodium tetraphenylborate with 0.005M cetylpyridinium chloride till the indicator turns blue.
- Repeat this without using potassium chloride.
Calculation:
The calculation formula is :
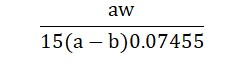
Where ‘a’ is the volume of 0.005 M cetylpyridinium chloride required without potassium chloride, ‘b’ is the volume required with potassium chloride, and ‘w’ is the weight of potassium chloride taken in grams.
Reference: This method is from the Indian Pharmacopoeia.
Read More:
- 0.1 M KOH Solution Preparation and Standardization
- 0.1 M Sodium Nitrite Preparation and Standardization
- 0.05 M EDTA Solution Preparation and Standardization
- Preparation and Standardization of 0.1 M Ceric Ammonium Sulphate
- Preparation and Standardization of 0.1 M Disodium Edetate (EDTA)
- How can I Prepare and Standardize 0.5 M Sulfuric acid?
- 1.0 M Sulfuric Acid Solution- Preparation, Standardization, Reagents, Formula
- 0.1 M Sodium Hydroxide (NaOH), Preparation and Standardization
- Preparation and Standardization of 1 M Sodium Hydroxide Solution (NaOH)
- Preparation and Standardization of 1.0 M Hydrochloric Acid
- Preparation and Standardization of 0.1 N HCl

