Change Control system SOP, including preparation of Format, Numbering, Initiation, Change & Assessment by QA, Review, Approval, Implementation, Distribution, Retrieval, Annexures, and closing of change control.

1.0 OBJECTIVE: To lay down the procedure for Change Control system SOP.
2.0 SCOPE: This Change Control system SOP is applicable to all related user departments as specified in section 5.5 (SOP Title: SOP of SOP), for handling and effecting the changes of approved documented procedure systems, facility, equipment, utility, software, and process as applicable in their department.
3.0 RESPONSIBILITY:
3.1 Originating Department for initiation of the proposed change and implementation of the approved change.
3.2 QA department for assessment, approval, and monitoring of the change.
3.3 Regulatory affairs department for review, approval of change ( as applicable), and intimation to registration authority.
4.0 ACCOUNTABILITY:
4.1 Head-Quality Assurance.
5.0 PROCEDURE for Change Control system SOP
5.1 Change control system gives the procedure for the following:
5.1.1 Initiation of Change Control
5.1.2 Review , Change & Assessment by QA
5.1.3 Final approval of change from QA
5.1.4 Monitoring and closing of change control
5.2 The ownership of the change is with the head of the originating department, who is responsible for initiating the change.
5.3 Identification of ownership for different changes and consent to be obtained from different departments is mentioned in change initiation matrix as per Appendix-I.
5.4 Part-I: INITIATION OF CHANGE CONTROL
5.4.1 The initiating department shall request QA to issue “Change control form”.
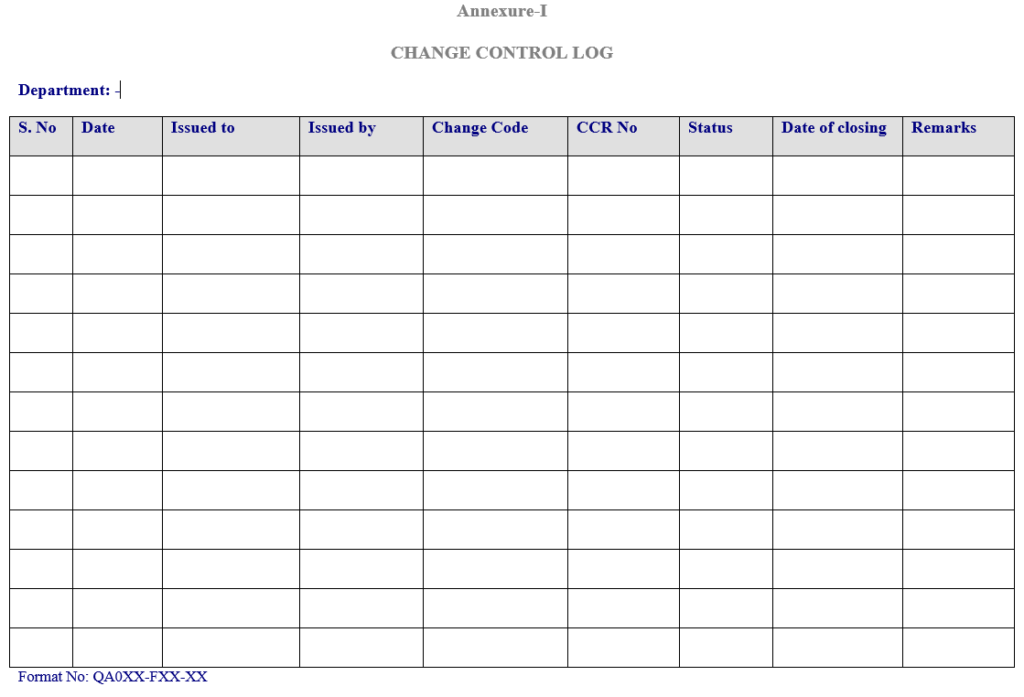
5.4.2 Document change control in charge to issue “Change control form” against the “Change control log“ which is maintained year-wise & department wise in QA as per Annexure-I.
5.4.3 Initiating department to furnish the details under section 3.0 as per Part-I under “change control form” (Annexure-II) (Refer Appendix-I).
5.4.4 Initiating department shall furnish the details of proposed change(s) as per section 3.0 of Part-I under “change control form”. Reason for change ( s) shall also be indicated as under section 4.0 of “change control form”, attach supporting documentation as applicable based on which change is proposed.
5.4.5 The nature of change i.e., temporary/permanent shall be indicated under section 6.0 of “Change control form”.
5.4.6 All changes proposed shall be authorized by respective initiating department head /his deputy.
5.4.7 The representative /Responsible person of the initiating department shall sign in the change initiated by column of Part-I in “Change Control form” (Annexure-II).
5.4.8 Head of the initiating department/deputy shall approve the proposed change and shall sign as authorized under section 10.0 of “Change control form” sign under Authorized by column of Part-I in Change Control Form.
5.5 Part-II A: CHANGE CONTROL CONSENT AND REVIEW
5.5.1 User department /Initiating department to submit the “Change control form” to Quality Assurance.
5.5.2 Quality assurance department to circulate the change control form to all the concerned departments and obtain their review comments ( the related departments, depending on the scope of change to refer appendix-I but are not limited and shall proceed on a case-by-case basis).
5.5.3 Responsible person of the department shall review the proposed change and record the review comments under section 11.0 of change control form.
5.5.4 The department head /deputy shall finally indicate the change acceptance and shall sign and date under section 11.0 of change control form.
5.6 Part-II A: CHANGE CONTROL ASSESSMENT BY QA
5.6.1 On receipt of change control form along with review comments of all other related departments, the document change control incharge of QA shall assess the change and based on the scope shall assign the change code as per Appendix-I.
5.6.2 QA shall assign the change control number as below:

5.6.3 Record the code number and change control number in the “Change control Log” ( Annexure-I).
5.6.4 Document change control incharge shall assess the level of change viz:
5.6.4.1 Type -A: Minor changes which do not have any detectable impact on the quality attributes of the product and having no regulatory impact.
5.6.4.2 Type -B: Changes that are likely or may have minor impact on the quality attributes of the product and requires intimation to registration agency.
5.6.4.3 Type -C: Changes that are having significant impact on the quality attributes of the product and require pre-approval from the registration agency.
5.6.5 Quality Assurance department to circulate the proposed “Change control form” to all the concerned department head and obtain their review comments.
5.6.6 QA shall also assess and review the impact of change on related documents under section no 13.0, Part-II of “Change control form”.
5.6.7 QA shall recommend the action to be carried out for the proposed change and documents under section no 14.0 , Part-II of “Change control form”.
5.6.8 The document change control in charge shall evaluate the impact of the proposed change and shall sign as change evaluated in the “Change control form”.
5.6.9 In case the change falling under the scope of Type-B or Type-C as per section no 12.0, sent the “Change control form” to Regulatory affairs for review and approval.
5.6.10 Regulatory affairs to review the effect of proposed change in the submitted DMF/ANDA/product Dossiers, other submitted customer documents etc.
5.6.11 Regulatory Affairs Department shall write comments for the regulatory submission requirements for the approval of the change.
5.6.12 If change require submission of pre-approval supplement, Regulatory Affairs will submit the same, and the change will be implemented after obtaining the regulatory approval.
5.6.13 In other case as applicable, regulatory affairs shall file Change being effected supplement or include the change in Annual Update.
5.6.14 Responsible person shall evaluate the impact of change and record his/her review comments under section 15.0 of “change control form”.
5.6.15 Head of regulatory affairs shall finally evaluate the change and shall approve the proposed change and sign in the “Change control form”.
5.7 Part-III: FINAL APPROVAL OF CHANGE CONTROL BY QA:
5.7.1 QA to evaluate the proposed change against the checklist as per section no 16.0, Part-III of “Change control form”.
5.7.2 Document change control in charge shall evaluate the action points mentioned under section 16.0 of “Change control form”..
5.7.3 Finally, Head-QA shall approve the proposed change and shall sign with date under section no 17.0 of “Change control form”.
5.8 Part-IV: MONITORING & CLOSING OF CHANGE CONTROL:
5.8.1 If the change proposed is approved by Head-QA, QA shall indicate and inform the initiating department for executing the proposed change.
5.8.2 Intimation shall be given if required, by calling for a meeting with the initiating department head and concerned department heads as applicable.
5.8.3 The approved “Change control form” shall be sent to the initiating department for executing the proposed change.
5.8.4 After the execution of the proposed change, the completed “Change control form” shall be returned to Quality Assurance for archival.
5.8.5 Document change control in charge shall monitor and ensure the completion of change and record under section no 18.0 of “Change control form”.
5.8.6 Closing of Change Control
5.8.6.1 On approval of change control, it is the responsibility of Quality Assurance to verify that whether the change is implemented as per the proposal, and verification shall be documented in section no 18.0 of the “Change control form” as per Annexure-II.
5.8.6.2 In case of temporary changes, QA shall ensure that old system is back in place after recommended period of change and close the change control.
5.8.6.3 Head-QA shall approve the closing of change control after ensuring the completion of change along with the other related requirement implementation as described under section 18.0 of “Change control form”.
5.9 Documentation change control in charge shall complete the “Change Control Log”(Annexure-I)
5.10 QA shall compile and archive all executed forms of Change control along with all supporting documents and file year-wise & department- wise.
6.0 ABBREVIATIONS
6.1 CCR-Change Control Request
6.2 DMF-Drug Master File
6.3 ANDA-Abbreviated New Drug Application
6.4 QA-Quality Assurance
7.0 ANNEXURE
7.1 Annexure-I-Change Control log
7.2 Annexure-II-Change Control form
END OF DOCUMENT

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].

