Founder | Pharmaceutical Professional
✓ Reviewed by: Pankaj Sharma - Quality Control Specialist
Reviewed for Quality Control accuracy, laboratory practices, and technical relevance
📅 Last Updated: May 15, 2026
1.0 Purpose: To describe the method for dispensing of active raw material from mother bottle to baby bottle as per requirement.
2.0 Aim: This Standard Operating Procedure is applicable to the distribution of active raw material supplied by approved vendor from mother bottle to small sterile bottles as required for formulation of a batch at production floor at Pharmaceuticals.

3.0 Responsibility:
3.1 Officers/ Executives shall perform activity.
3.2 Head production/Incharge shall be responsible for control of the activity & implementation of this SOP.
3.3 Head, QA/designee shall be the authorizing authority of the SOP.
4.0 Safety Consideration:
4.1 Wear the sterile gowning (Goggle, Gloves, Hood, booties, Gown, and Mask) during the handling of active raw materials.
4.2 Person handling the bulk should be immunized for the particular vaccine.
4.3 Aseptic dispensing activity shell to be performed in class A area surrounded by class B.
5.0 Equipments:
5.1 Magnetic stirrer
5.2 Weighing balance
5.3 Cold room (2 – 8 degree C )
5.4 Deep freeze ( -20 degree C )
5.5 Dispensing Booth
6.0 Materials and reagents:
6.1 70% sterile Isopropyl alcohol.
6.2 Pre-sterile bottle.
6.3 Sterile measuring cylinder.
6.4 Clean room mop.
6.5 Scissors.
6.6 Waste bin.
6.7 Arterial clamps.
6.8 Pinch crock.
6.9 Calibrated Sterile glass bottle with cap.
6.10 Sterile glass bottle with SS siphon.
7.0 Preparation of solutions:
Not Applicable
8.0 Procedure:
8.1 Mother bottle is the container with bulk of active raw material, which is supplied by approved vendor.
8.2 Baby bottle is the container with sterile dispensed active raw material from mother bottle as per batch requirement.
8.3 Transfer of mother bottle from warehouse to production as per SOP (Transfer and Receipt of bulk (ARM) from Warehouse to Production area).
8.4 Check the status of mother bottle to be dispensed in presence of IPQA personnel for followings:
8.4.1 QC release status.
8.4.2 Expiry date/Retest date.
8.4.3 Total quantity.
8.4.4 Conc. / COA of the ARM.
8.5 Calculate the active raw material requirement as mentioned in different product’s SOP for formulation of various products.
8.6 Stores issue the mother bottle to production for dispensing into small sterile bottles as per requirement.
8.7 Receive the mother bottle from warehouse as mentioned in Bulk ARM Transfer SOP.
8.8 Clean the outer surface of bottles with filtered 70 % IPA before its use.
8.9 Check the sterilization status of sterilized material to be used for dispensing.
8.10 Clean & put on the dispensing booth 30min as per cleaning SOP before starting of ARM dispensing activity.
8.11 In case of active raw material stored below 0°C, the ARM is to be thawed at 2- 8 ºC for 24- 48 hrs before dispensing.
8.12 Ensure cleaning & operating status of the area & dispensing booth prior to dispensing as per cleaning SOP & take line clearance for start of dispensing process.
8.13 Ensure the bottles into which the ARM is to be dispensed are sterilized with in last 48 hours and are intact.
8.14 Open the sterile glass bottles inside the dispensing booth, which are packed in sterile packing.
8.15 Insert sterile S.S. siphon aseptically into the mother bottle and tight the cap properly.
8.16 Dispense the calculated amount of bulk for a standard batch size by transferring the ARM from mother bottle into the sterile baby bottle aseptically by using a peristaltic pump.
8.17 After transferring, replace the s.s. siphon from mother and baby bottle with sterile cap and tightly cover.
8.18 ARM shall be dispensed by rounding off the calculated quantity up to tenth digit eg.
If calculated quantity comes to be 182.5 ml, then ARM to be dispensed shall be 190 ml.
8.19 During dispensing of whole cell pertussis, the mother bottle needs to be stirred continuously overnight to get homogenous bulk dispensed into the baby bottles.
8.20 No two mother bottle bulk should be mixed in a single baby bottle while dispensing.
8.21 Attach Status label on baby bottle with label containing name of API, batch no., baby bottle no., and date of dispensing, duly signed by responsible persons.
8.22 A duplicate label is tagged onto the bottle so that label can be attached in the BPR of respective batch where the bulk is to be used.
8.23 After completing the dispensing seal, the cap of mother and baby bottle should be secured with paraffin tape properly.

Dispensed baby bottles are numbered as AR-MAF/XXXXX / YY – ZZZ
Whereas:- AR – Abbreviations of active raw material
MAF – Abbreviation of manufacturer
XXXXX – Batch No. of active raw material
YY – Mother Bottle No. (Container No.)
ZZZ – Dispensed Bottle No. starting from 001.
*The number of digits depends on the name of ARM coding in annexure -3 of this SOP.
8.24 Mother bottle number. (Container no.) Provided by warehouse shall be considered for numbering the dispensed baby bottle number.
8.25 Dispensed Baby bottle no. shall start from 001 for the given manufacture’s batch no. and container no. When container number of the same manufacture’s batch is getting changed, the no. of Dispensed Baby bottle shall again start from 001. e.g Whole cell Hepatitis B bulk from manufacture. BioGen Pharma, batch no. HBX214, mother bottle no.(container no.) 03, the numbering system for dispensed Baby bottle shall be AR-MAF/HBX214/03-001, and similarly for mother bottle no.(container no.) 04 of the same manufacture’s batch shall be AR-MAF/HBX214/04-001.
8.26 Perform the environmental monitoring during dispensing as per SOP (Monitoring and Recording Critical Environmental Parameters of Different classes). Attached the EMP reports and material sterilization data.
8.27 Store the dispensed baby bottles as per manufacturer’s recommendations in a cold room until its use.
8.28 Before the day of formulation, SS siphons shall be sterilized with respective batch formulation load.
8.29 Sterilized S.S. siphons shall be inserted into dispensed baby bottles before it’s used in batch formulation. (Perform the aseptic operation as per SOP for Aseptic Handling, Operation)
8.30 Clean the area of dispensing booth as per cleaning SOP & send all used material for decontamination after use as per SOP Decontamination of used Material Contacted with API.
9.0 Reporting:
9.1 Dispensing record of active raw material from mother bottle into smaller bottles shall be recorded in annexure -1& usage of ARM in annexure -2.
10.0 References:
10.1 Media fill document
11.0 Annexure:
Attached.
12.0 Distribution:
| Master Copy | Archived with Quality Assurance Department |
| Controlled Copy 1 | Production Department |
| Subsequent controlled copies | Department (s) making requisition |
13.0 History:
| Effective Date | Status | Changes/Reasons for revision |
| Revision No : 00 | ||
| Revision No : 01 | ||
| Revision No: 02 | ||
| Revision No: 03 |
14.0 Abbreviations:
14.1 HOD: Head of the Department
14.2 ID No. : Identification number
14.3 SOP: Standard Operating Procedure
14.4 VFP: Vaccine Formulation Plant
14.5 DISPENSING BOOTH: Laminar air flow
14.6 VDISPENSING BOOTH: Vertical laminar air flow
14.7 IPA: Isopropyl alcohol
14.8 NMT: Not more than
14.9 ARM: Active raw material
14.10 EMP: Environmental monitoring plan
14.11 Q.C.: Quality control
14.12 S.S: Stainless steel
14.13 N/A : Not applicable
Annexure-I
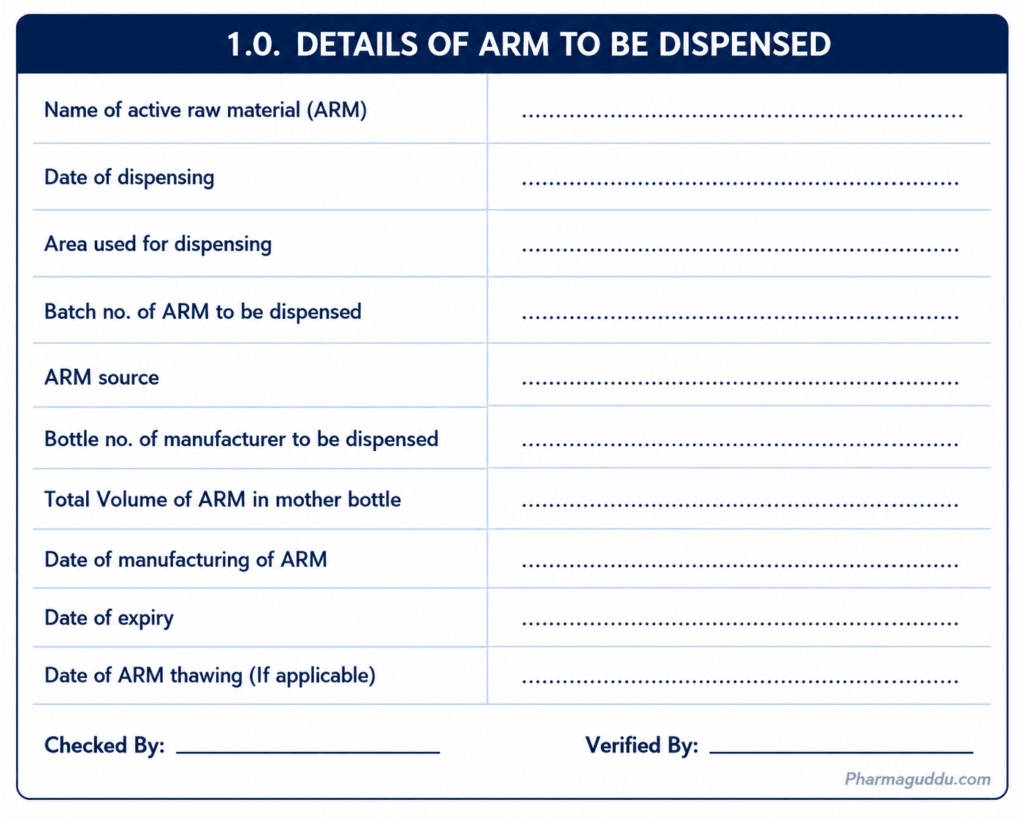
3.0 DISPENSING OF ARM
3.1. Preparation for Dispensing of ARM:
Wash the material required for dispensing as per the current version of SOPs.
3.1.1. Washing & Material Preparation Details for Dispensing:
Date of washing & preparation of materials: …………………………………
Washed by…………………… Checked by………………………
Date………………………… Date………………………………
3.1.2. Result of water rinse after washing:
| Date of sampling | Name of sample | No. of samples | Result | Checked by |
| Pass/ Fails | ||||
| Pass/ Fails |
Verified by ……………………………. Sign: …………………… Date: ……………
3.1.3. Material Sterilization Details:
Date of sterilization : …………………………………
Equipment I.D. of Autoclave: …………………………………
Load no. : …………………………………
Autoclave Run Details:
| Cycle no. | Description | Sterilization Hold time | Done By | Checked By | |
| From | To | ||||
* Attach checked strip chart for the above run: Attached /Not attached
3.2. Dispensing details:
a. Date of dispensing : ………………………………………….
b. Room no used for dispensing : ……………………………………………
c. EQ.I.D. of DISPENSING BOOTH used : ………………………………………………
3.2.1. Checklist for ARM dispensing:
| S.no | Check points | Observations |
| 1. | Check the COA of the received ARM to be dispensed. | |
| 2 | Check calculations of ARM required to be dispensed. | |
| 3 | Packing of ARM received & ensure the bottles are properly sealed before dispensing | |
| 4 | Details of ARM on Label of the bottle match with the COA. |
Checked by:…………Verified by:…………..
3.2.2. Line Clearance for ARM Dispensing:
| S.no. | Description of the checkpoints | SOP Ref. no. | Remarks |
| 1 | Area has been cleaned & disinfected | ||
| 2 | DISPENSING BOOTH cabinet cleaned & put on before 30 min of dispensing | ||
| 3 | Presence of all materials required for dispensing | ||
| 4 | All the material used has been sterilized. | ||
| 5 | Temperature & humidity of the dispensing area was checked & found ok |
Checked by:………………………….. Verified by: …………………………
Date: Date:
3.3. Thawing of ARM
Active raw material after receipt from stores is to be thawed in cold room at temperature 2-8 °C.
| Thawing Start time/date | Thawing End time/date | Total time | Done by. | Checked by |
* Attach checked raw data for temperature profile during thawing.: Attached /Not Attached
3.4. Environmental monitoring of Formulation & Blending Area during ARM dispensing :
a) Settle plate : QC Report no:.. ………………………..
b) Volumetric air sampling : QC Report no: ………………………….
c) Personnel monitoring : QC Report no: ………………………….
d) Surface sampling : QC Report no: ………………………….
e) Particle count : ………………………………………
Checked by:………… Checked by:……….. Verified by:…………
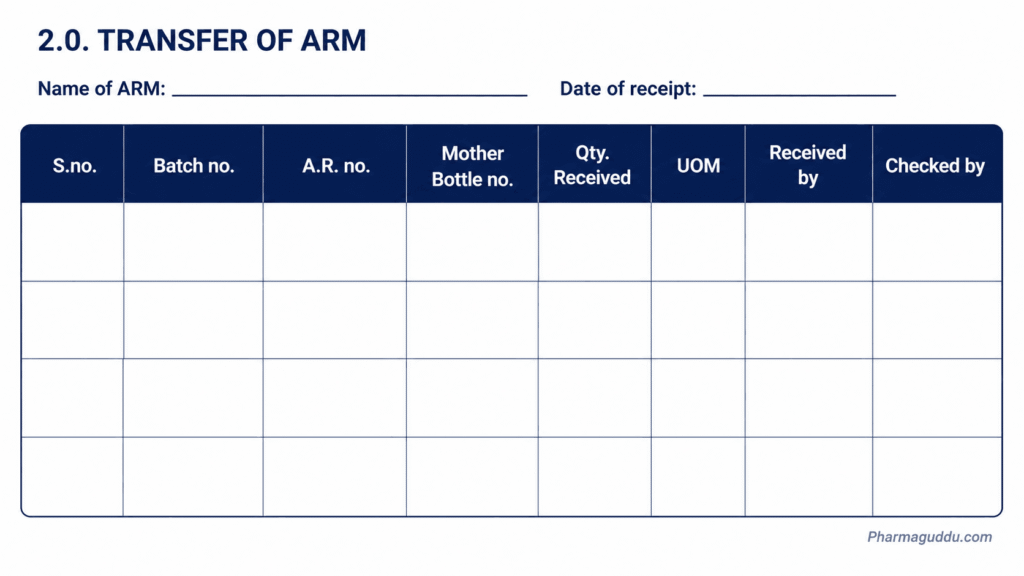
3.5. Dispensing of ARM
Name of ARM: ………………………………………………….. Date of dispensing:………………
| Bulk Mother Bottle No. | Batch no. of ARM | Dispensed Bottle no. | UOM | Qty. dispensed | Volume Dispensed by | Volume checked by |
3.5.1. The distributed bulk bottles are stored in cold room / deep freeze until use:
EQ.ID. of cold room / deep freeze : …………………………………………………………….
Location of cold room / deep freeze :…………………………………………………………….
Transferred by: ………………………………….. Verified by:………………………………….
(Warehouse) (QA)
Monitored the temperature of the cold room or deep freeze and record the temperature.
4.0. POST DISPENSING ACTIVITY:
4.1. Decontamination and disposal of used materials:
1) Materials used for dispensing are to be decontaminated as per SOP.
2) Reusable material to be washed as per SOP after decontamination and reused.
3) Disposable material to be disposed as per SOP Handling Scrap and Disposal after decontamination.
Date of decontamination:…………………………………………….
Done by:…………………………… Checked by:………………………………………
4.2. Post-dispensing Cleaning:
After formulation, area & vessels are cleaned & materials used are to be disinfected and disposed.
Check points after formulation:
| Sr. No. | Actitvity | Remark |
| 1. | Check for leftover used glass bottles | (Done / Not Done) |
| 2. | Check for empty bulk bottle that has been sent for disinfection | (Done / Not Done) |
| 3. | Check for cleanliness of work benches & area | (Done / Not Done) |
Date of cleaning : …………………………
Cleaned by : …………………………. Checked by : ……………………………

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].

