Founder | Pharmaceutical Professional
✓ Reviewed by: Pankaj Sharma - Quality Control Specialist
Reviewed for Quality Control accuracy, laboratory practices, and technical relevance
📅 Last Updated: May 16, 2026
1.0 Scope: Procedure is applicable to perform the colour test of the sample wherever specified.
Learn: Procedure to Check Clarity of Sample Solution
2.0 Apparatus:
You will need:
- Standard test tubes
- 100 ml volumetric flask
- 25 ml measuring cylinder
- 10 ml graduated pipette
- Flat-bottomed color-matching test tubes
3.0 Reagents:
Use analytical grade (AR) chemicals for the following:
- Ferric chloride hexahydrate
- Cobaltous chloride
- Cupric sulphate
- Concentrated hydrochloric acid
- Potassium iodide
- Starch solution
- 0.1 M sodium thiosulphate
- Hydrogen peroxide (10 volumes)
- Sodium hydroxide solution
- Dilute sulphuric acid
- Acetic acid

4.0 Method:
A. Ferric Chloride Colorimetric Solution (FCS)
Dissolve roughly 55 g of ferric chloride hexahydrate in a mixture of 25 ml hydrochloric acid and 975 ml distilled water. Bring the total volume to 1000 ml.
Standardization:
- Pipette 10 ml of the FCS into a 250 ml iodine flask.
- Add 15 ml distilled water, 3 g potassium iodide, and 5 ml hydrochloric acid.
- Let the mixture sit for 15 minutes.
- Add 100 ml distilled water.
- Titrate the released iodine with 0.1 M sodium thiosulphate. Add 0.5 ml starch solution near the end of the titration. It acts as your indicator.
- Run a blank test. Adjust your final numbers if needed.
Note: 1 ml of 0.1 M sodium thiosulphate equals 0.02703 g of FeCl₃·6H₂O. Adjust the final volume with more of the acid-water mixture until every milliliter contains exactly 0.045 g of FeCl₃·6H₂O. Store this solution away from light.
B. Cobaltous Chloride Colorimetric Solution (CCS)
Dissolve about 65 g of cobaltous chloride in 25 ml hydrochloric acid and 975 ml distilled water. Bring the volume to 1000 ml.
Standardization:
- Pipette 5 ml into a 250 ml iodine flask.
- Add 5 ml hydrogen peroxide and 15 ml sodium hydroxide solution.
- Boil for 10 minutes. Let it cool.
- Add 2 g potassium iodide and 60 ml dilute sulphuric acid.
- Gently shake to dissolve any solid that forms.
- Titrate the released iodine with 0.1 M sodium thiosulphate. Add 0.5 ml starch solution as the liquid nears the pink end-point.
- Run a blank test. Adjust your numbers if needed.
Note: 1 ml of 0.1 M sodium thiosulphate equals 0.02379 g of CoCl₂·6H₂O. Adjust the final volume with the acid-water mixture until every milliliter contains 0.0595 g of CoCl₂·6H₂O.
C. Cupric Sulphate Colorimetric Solution (CSS)
Dissolve roughly 65 g of cupric sulphate in 25 ml hydrochloric acid and 975 ml distilled water. Bring the volume to 1000 ml.
Standardization:
- Pipette 10 ml into a 250 ml iodine flask.
- Add 40 ml distilled water, 4 ml acetic acid, 3 g potassium iodide, and 5 ml hydrochloric acid.
- Titrate the released iodine with 0.1 M sodium thiosulphate. Add 0.5 ml starch solution as the liquid turns pale brown.
- Run a blank test. Adjust your numbers if needed.
Note: 1 ml of 0.1 M sodium thiosulphate equals 0.02497 g of CuSO₄·5H₂O. Adjust the final volume with the acid-water mixture until every milliliter holds 0.0624 g of CuSO₄·5H₂O.
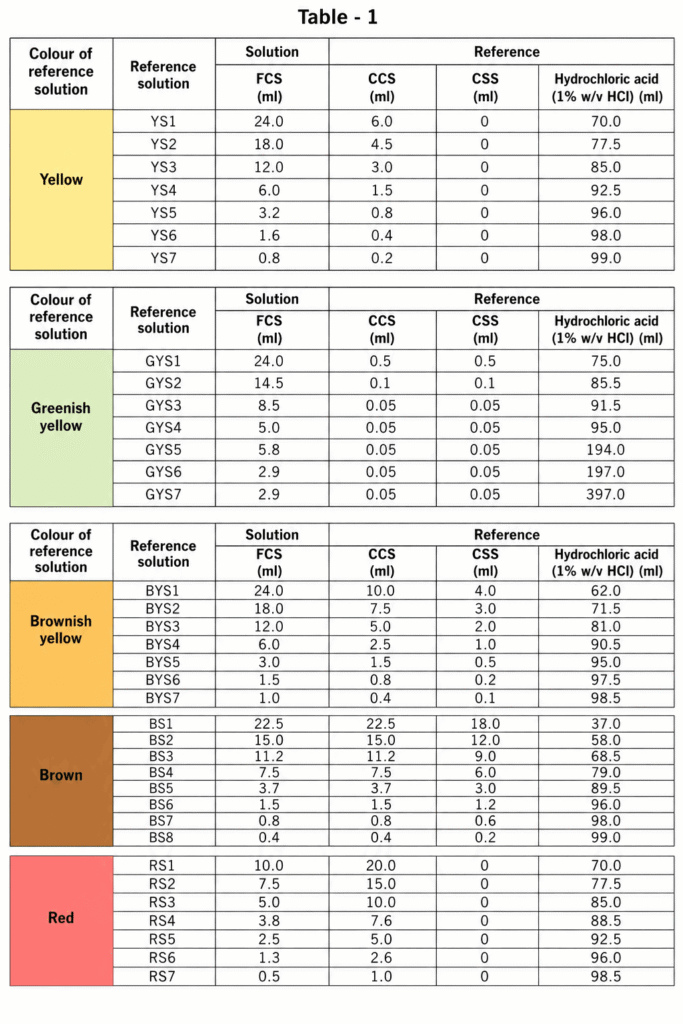
Reference Solution:
Mix the prepared colorimetric solutions with 1% w/v hydrochloric acid. Follow the exact ratios listed in Table 1. Always prepare reference solutions right before use. The stock colorimetric solutions can be stored for later use.
Testing Procedure:
- Use a flat-bottomed neutral glass test tube (15 to 25 mm wide).
- Pour in enough sample solution to fill the tube to a 40 mm depth.
- Take a second matching tube. Fill it with the same volume of water, the original solvent, or the specified reference solution.
- Look straight down into both tubes. Use diffused light and place them against a white background. Compare the colors.
Colourless Solution:
A solution is considered colourless if it matches water or its solvent. It also passes if it does not appear darker than reference solution BS8.
5.0 Reference: IP-1996 (Appendix 6, page A-78)

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].

