Founder | Pharmaceutical Professional
✓ Reviewed by: Pankaj Sharma - Quality Control Specialist
Reviewed for Quality Control accuracy, laboratory practices, and technical relevance
📅 Last Updated: April 27, 2026
1.0 OBJECTIVE: Cleaning Validation Protocol for Sifter to ensure that the cleaning procedure for Sifter removes residues to the extent of compliance with pre-determined acceptance level.
2.0 SCOPE: This protocol applies to Pharmaceutical Manufacturing Facility (name of company and place)
3.0 RESPONSIBILITY: It is a joint responsibility of Quality Control, Production and Quality Assurance departments. The detailed responsibility matrix is given below:
- Protocol Preparation – Executive QA
- Protocol Approval – Head – QA
- Protocol Execution – Head – Production
- Sampling as per Protocol – Executive – QA
- Testing as per Protocol – Executive – QC
- Test Results Review and Approval – Manager – QC
- Validation Data Compilation – Executive – QA
- Validation Final Approval – Head – QA

4.0 PROCEDURE: for Cleaning Validation Protocol for Sifter
- Clean the equipment as per SOP (Title: Procedure for cleaning of Sifter).
- Sample from the specified sampling points (6.0) as per the sampling plan (5.0).
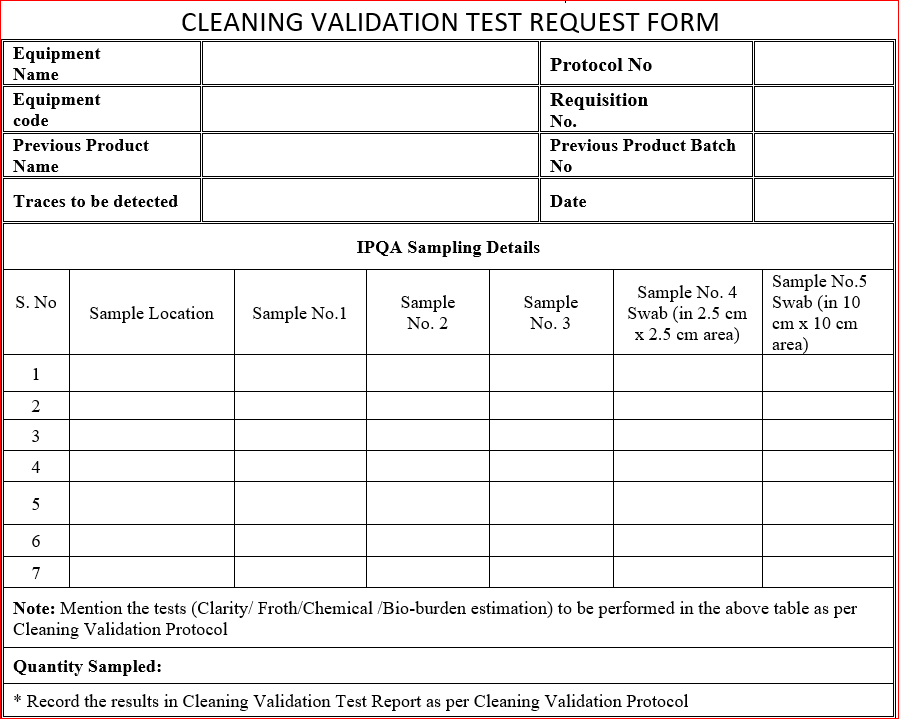
5.0 SAMPLING PLAN
- Sample from first rinse (after washing with raw water).
- Sample from second rinse (after cleaning with 0.1% liquid soap solution and raw water).
- Sample from final rinse of purified water.
- Swab (in 2.5 cm x 2.5 cm area) for chemical analysis.
- Swab (in 10 cm x 10 cm area) for bio-burden.
6.0 SAMPLING POINTS
- Upper Hopper
- Sieve used for the batch
- Powder discharge assembly
Note: Please refer to ANNEXURE – B
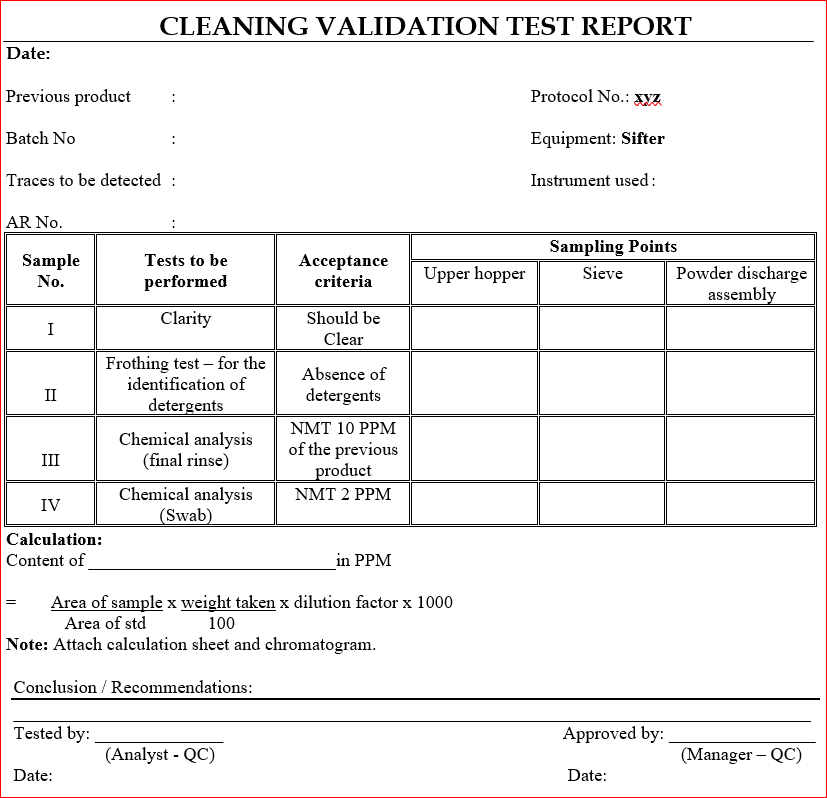
7.0 TEST / METHOD
- Inspect visually for the absence of previous product in the equipment (sample I).
- Determine the absence of detergents by froth method (sample II).
- Determine the concentration of active ingredient of the previous product in the final rinse (sample III).
- Determine the concentration of active ingredient of the previous product in the swab (sample IV).
- Report and record the above test (1,2,3 and 4) results in ANNEXURE – C.
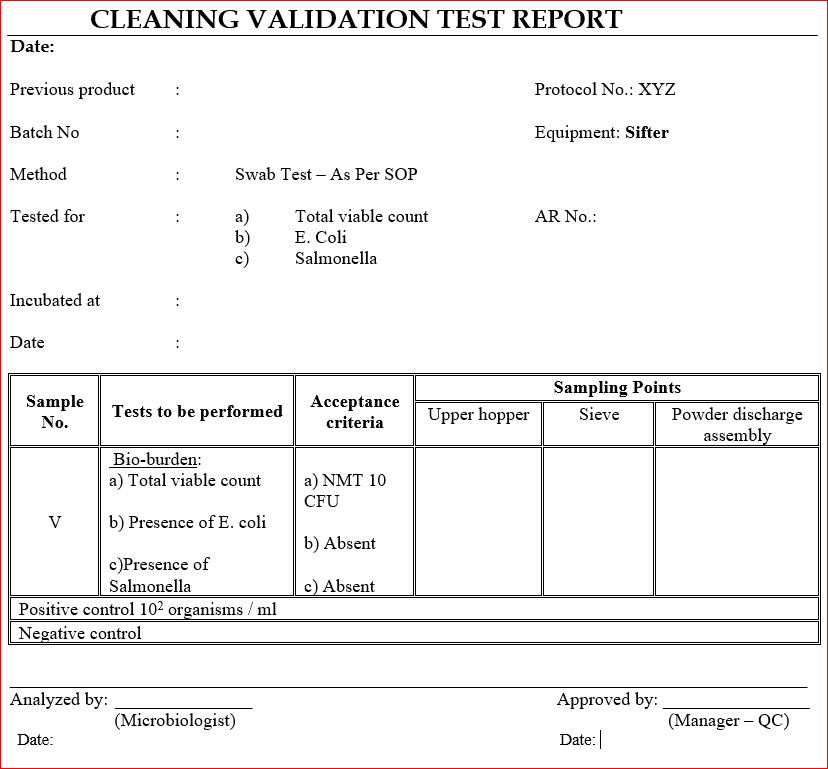
- Determine the bio-burden (sample V) and record the results in ANNEXURE – D.
8.0 ACCEPTANCE CRITERIA
- No traces of the powder shall be visible on any part of the machine (sample I).
- Detergent should not be identifiable by frothing test (sample II).
- Concentration of the previous product should not be more than 10 PPM in the final rinse (sample III).
- Concentration of the previous product should not be more than 2 PPM in the swab (sample IV).
- Bio-burden limits are as below (sample V):
- Total CFU – NMT 10 CFU
- E. Coli – absent
- Salmonella – absent
9.0 ANNEXURE – A
10.0 ANNEXURE – B
SCHEMATIC SKETCH OF SAMPLING POINTS

11.0 ANNEXURE – C
12.0 ANNEXURE – D

Naresh Bhakar is the Founder and Author at Pharmaguddu.com, bringing his extensive expertise in the field of pharmaceuticals to readers worldwide. He has experience in Pharma manufacturing and has worked with top Pharmaceuticals. He has rich knowledge and provides valuable insights and data through his articles and content on Pharmaguddu.com. For further inquiries or collaborations, please don’t hesitate to reach out via email at [email protected].

