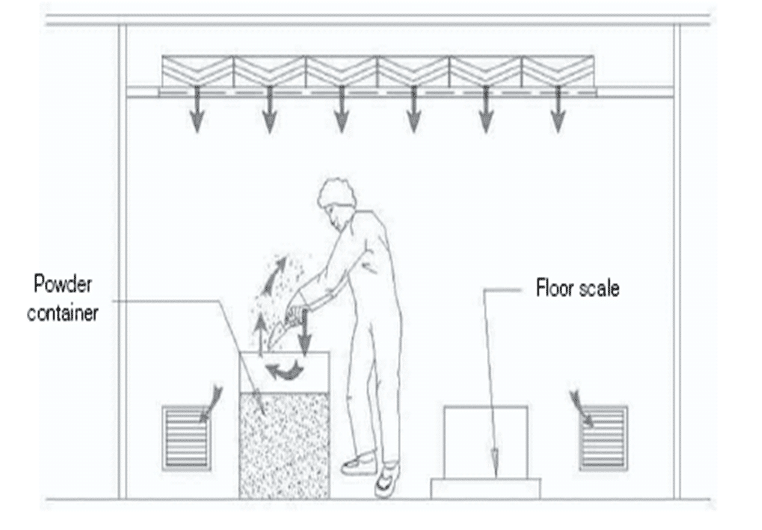
SOP for Cleaning and Operation of vials washing machine
Learn about the Standard operating procedure on the vials washing machine in pharmaceuticals along with their Objective, Scope, Responsibility, procedure, precautions, cleaning, and acceptance criteria. 1.0 Objective: To lay down a procedure for Cleaning and operation of vials washing machine to make vials free from particulate matter.2.0 Scope: This SOP applies to the Injection Section of the production department. … Read more