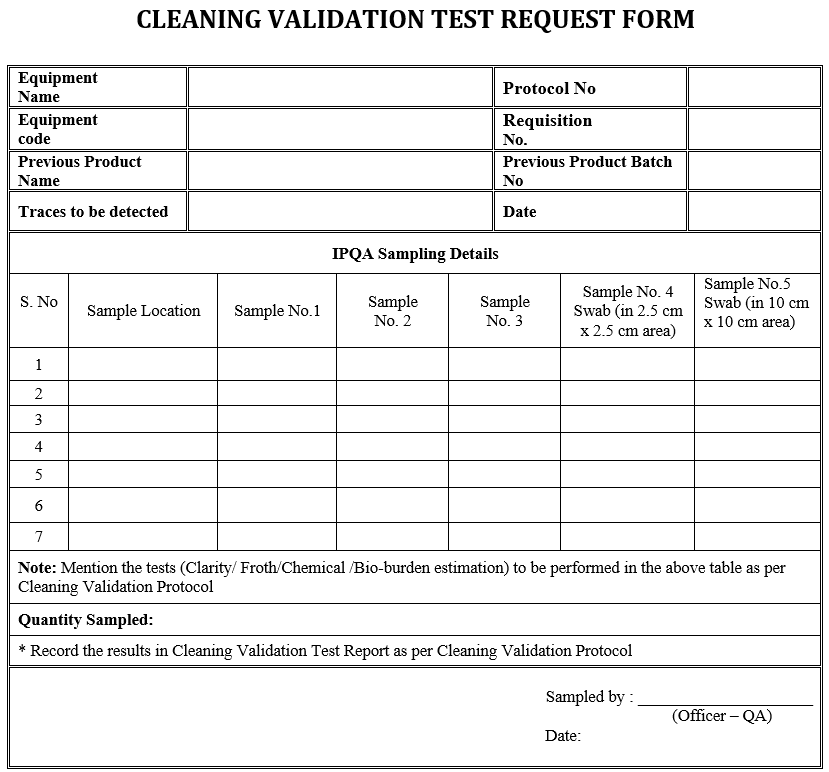
Cleaning Validation Protocol for Multi mill
1.0 OBJECTIVE: Cleaning Validation Protocol for Multi Mill is designed to ensure that the cleaning procedure for multi mill removes residues to the extent of compliance with pre-determined acceptance level. 2.0 SCOPE: This protocol applies to Pharmaceutical Manufacturing Facility. 3.0 RESPONSIBILITY: It is a joint responsibility of Quality Control, Production, and Quality Assurance departments. The … Read more